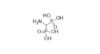
This foundational α-aminobisphosphonic acid features a unique methylene-bridged geminal bisphosphonate structure with a primary amino group directly attached to the central carbon — H₂N-CH(PO₃H₂)₂. It serves as the parent scaffold for a diverse family of N-substituted aminomethylenebisphosphonates, renowned in medicinal and agricultural chemistry.
Here are representative molecular structure visuals of (aminomethylene)bisphosphonic acid and closely related aminobisphosphonates:
Key Features & Properties
- Strong chelating agent — gem-bisphosphonic acid groups provide high affinity for metal ions (especially Ca²⁺, Mg²⁺, and transition metals)
- Highly polar and water-soluble — excellent for aqueous applications and biological systems
- Acidic nature — four ionizable protons (pKa values in the acidic to neutral range), enabling multiple salt forms
- White to off-white crystalline solid (free acid form); hygroscopic and typically handled/stored under controlled conditions
- Stable under normal conditions but decomposes at elevated temperatures
Prime Applications & Research Interest
- Precursor for bioactive derivatives — N-substitution (e.g., pyridyl, phenyl, alkyl) yields potent herbicidal compounds that inhibit key plant enzymes like DAHP synthase (shikimate pathway) and glutamine synthetase, rivaling glyphosate in activity
- Bone-targeting & medicinal chemistry — aminobisphosphonate motif shows promise for bone affinity, similar to clinically used bisphosphonates; used in conjugates for drug delivery to hydroxyapatite/bone surfaces
- Enzyme inhibition studies — derivatives inhibit human P5C reductase (proline biosynthesis), showing antiproliferative effects against proline-dependent tumor cells
- Coordination chemistry & materials — building block for metal-organic frameworks, hybrid materials, and supramolecular assemblies
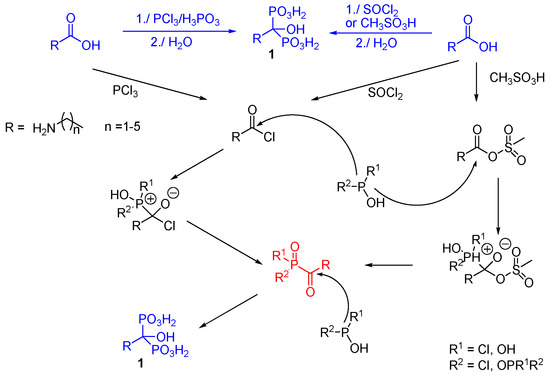
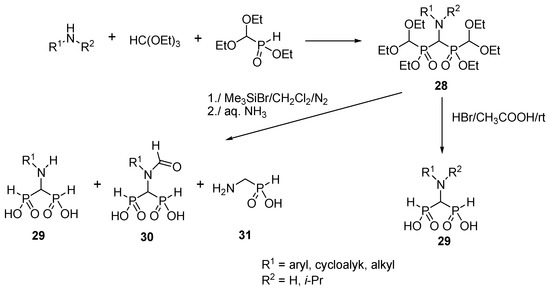
- Synthetic intermediate — versatile for three-component condensations and phosphorylation reactions to access complex aminobisphosphonates and bisphosphine oxides
Storage & Handling Recommendation: Store in a cool, dry place under inert atmosphere or as a stable salt. Use glovebox/Schlenk techniques for air-sensitive manipulations. Avoid prolonged exposure to moisture.
Explore the parent structure that inspired generations of potent enzyme inhibitors, bone-seeking agents, and next-generation agrochemicals — a cornerstone in modern phosphonate chemistry!


![561-41-1 α,α-Bis[4-(dimethylamino)phenyl]-4-(methylamino)-benzenemethanol](https://synovionindustries.com/wp-content/uploads/2025/12/561-41-1-1-300x175.jpg)
Reviews
There are no reviews yet.