Pentamethylcyclopentadienylbis(triphenylphosphine)ruthenium(II) chloride is a highly versatile, air-stable organoruthenium(II) half-sandwich complex featuring a pentamethylcyclopentadienyl (Cp*) ligand, two triphenylphosphine (PPh₃) ligands, and a chloride ligand coordinated to the ruthenium center. This electron-rich, coordinatively unsaturated species exhibits excellent stability and solubility in common organic solvents such as chloroform, dichloromethane, tetrahydrofuran, and acetone. It serves as a powerful precatalyst or direct catalyst in advanced organometallic transformations.
Key Features and Benefits:
- Exceptional Regioselectivity in Click Chemistry: Catalyzes the azide-alkyne cycloaddition (Huisgen reaction) to yield exclusively 1,5-disubstituted-1,2,3-triazoles — in contrast to the 1,4-regioselectivity of classic Cu(I) catalysis. It uniquely tolerates internal alkynes, expanding synthetic scope for triazole-based materials, bioconjugates, and pharmaceuticals.
- Living Radical Polymerization (ATRP/RAFT-like): Highly effective for controlled/living radical polymerization of methacrylates (e.g., MMA), acrylates (e.g., MA), and styrene, producing polymers with narrow molecular weight distributions (Mₙ/Mₙ < 1.1–1.2) and predictable chain growth.
- Robust and Tunable: The bulky Cp* ligand enhances thermal stability and electron donation, while the labile chloride and phosphine ligands allow easy activation/substitution for diverse catalytic cycles.
- High Purity Grade: Supplied as ≥95–98% pure for precise research applications in catalysis and synthesis.
Physical and Chemical Properties:
- Appearance: Yellow-brown to orange solid (powder or crystalline)
- Form: Solid
- Melting Point: Approximately 128–183 °C (decomposition reported in some sources)
- Solubility: Soluble in chloroform, dichloromethane, THF, acetone; insoluble in water
- Storage Conditions: Store at 2–8°C in a tightly sealed container, protected from moisture and light (air-stable under normal handling)
Applications:
- Regioselective Ru-catalyzed azide-alkyne cycloaddition (RuAAC) for 1,5-triazoles in drug discovery, materials science, and bioconjugation
- Living/controlled radical polymerization of (meth)acrylates and styrene for advanced polymers and block copolymers
- Precursor for other Cp*Ru(II) complexes via ligand substitution
- Research in organometallic catalysis, click chemistry, polymer chemistry, and homogeneous catalysis
This premium ruthenium complex is an indispensable tool for synthetic chemists seeking regiochemical control, controlled polymerization, or novel organometallic reactivity. For bulk quantities, custom synthesis, or certificates of analysis, contact our sales team.
Here are representative images of the chemical structure and typical appearance:
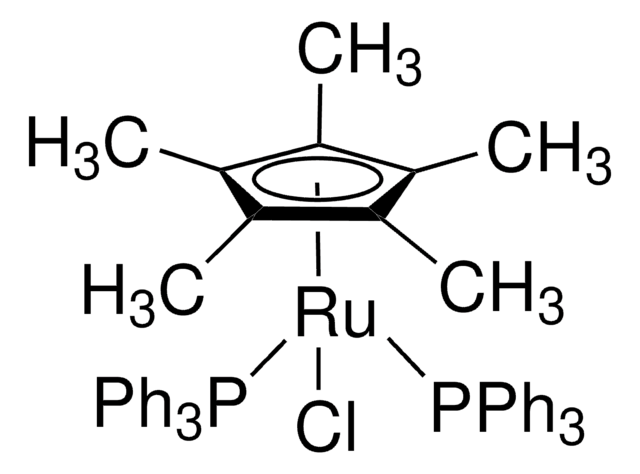


bis(3-bromopyridine)ruthenium(II)](https://synovionindustries.com/wp-content/uploads/2026/01/900169-53-1-300x163.jpg)
Reviews
There are no reviews yet.