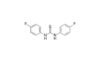
1,3-Bis(4-fluorophenyl)thiourea is a high-purity, symmetric diaryl thiourea compound featuring two para-fluorophenyl groups attached to the thiourea core. The incorporation of fluorine atoms enhances its electronic properties, reactivity, and potential biological interactions, making it a versatile building block in advanced organic synthesis and materials research.
Key Features and Benefits:
- Hydrogen-Bonding Capability: Acts as a bifunctional hydrogen-bond donor, ideal for organocatalysis and anion recognition.
- Catalytic Applications: Serves as an efficient ligand in metal-catalyzed reactions, such as copper(I)-supported catalysis for azide-alkyne cycloaddition (click chemistry) to form 1,2,3-triazoles.
- Reagent in Synthesis: Valuable intermediate for constructing heterocyclic compounds, including pharmaceuticals and agrochemicals.
- Biological Potential: Thiourea derivatives like this exhibit promising antimicrobial, antifungal, anticancer, and enzyme-inhibitory activities due to the thiourea scaffold and fluorine substitution.
- Coordination Chemistry: Forms stable complexes with transition metals, useful in coordination compounds and materials science.
- High Purity and Stability: Supplied in solid form, stable when stored sealed at room temperature.
Applications:
- Organocatalysis and asymmetric synthesis
- Pharmaceutical intermediates (e.g., for anticancer and antimicrobial agents)
- Agrochemical development
- Click chemistry and triazole synthesis
- Ligand in metal complexes for catalysis
- Research in medicinal chemistry and materials science
This fluorinated thiourea derivative is an essential tool for researchers seeking enhanced reactivity and selectivity in synthetic and biological applications.

![561-41-1 α,α-Bis[4-(dimethylamino)phenyl]-4-(methylamino)-benzenemethanol](https://synovionindustries.com/wp-content/uploads/2025/12/561-41-1-1-300x175.jpg)
Reviews
There are no reviews yet.